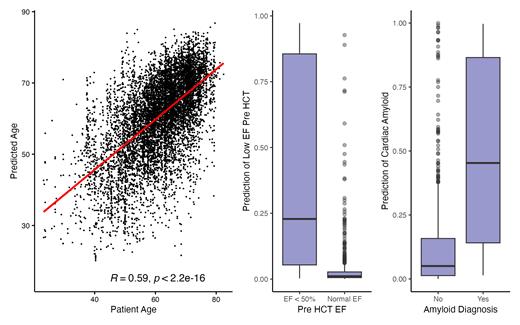
High dose chemotherapy followed by autologous hematopoietic cell transplantation (auto HCT) is a standard therapy for patients with plasma cell disorders (PCD). A barrier to improving candidate selection for auto HCT is appraisal of risk for transplant related complications. Electrocardiography (ECG) is a standard pre-HCT test to evaluate the potential for arrhythmia and structural heart disease. We hypothesized that an artificial intelligence (AI) based interpretation of ECG waveform data (AI-ECG) could enhance risk prediction for cardiac-specific and other outcomes following auto HCT for patients with PCD. To test this hypothesis, we used several AI-ECG tools developed using a deep-learning based approach that were trained to predict comorbidity (Attia et al, Lancet 2019 & Grogan et al, Mayo Clin Proc 2021). Model outputs include a scaled prediction value indicating increasing risk for atrial fibrillation/flutter (AF), reduced ejection fraction (EF), or cardiac amyloidosis. We evaluated this tool in 9,225 ECGs obtained from 849 patients (median 6 ECGs/patient, inter-quartile range: 3-12) who underwent first or subsequent auto HCT at Memorial Sloan Kettering Cancer Center between 2016-2022 and who had a diagnosis of multiple myeloma (N = 810), amyloidosis (N = 37), or other PCD (N = 2). The endpoints were to determine whether baseline AI-ECG prediction of AF risk correlated with peri-transplant AF events, and to explore whether model outputs correlated with survival and other cardiac clinical outcomes. The mean age of patients was 61.0 years (range 23.7 - 81.2) and median follow up was 2.7 years post auto HCT. Contributing cardiovascular risk factors included a prior history of AF (N = 101, 11.9%), coronary or cerebrovascular disease (N = 95, 11.2%), and diabetes mellitus (N = 98, 11.5%). Reduced dose melphalan (< 200 mg/m 2) was administered in 26%. The HCT-comorbidity index (HCT-CI) was high risk (≥3) in 52.7%. The one-year overall survival and progression-free survival in the whole cohort was 94% (95% confidence interval (95% CI): 92 - 95%) and 86% (84 - 89%), respectively. Among patients not in AF at pre HCT evaluation, the 90-day cumulative incidence of AF was 5.6% (4.2 - 7.3%). In univariable competing risk regression, baseline pre HCT AI-ECG appraisal of AF risk was strongly correlated with incidence AF in patients who were in sinus rhythm before HCT (hazard ratio (HR): 25.1, 95% CI: 11.7 - 53.7, p < 0.001). In a multivariable model adjusting for patient age and HCT-CI, AI-ECG AF predicted risk remained associated with incidence of post HCT AF (HR: 13.4, 95% CI: 5.9 - 30.4, p < 0.001). Considering a decision threshold identified during original model training, AI-ECG predicted AF events with an accuracy of 75% (Mcnemar's p < 2 x 10 -16). The precision, recall, and F1-score were 0.94, 0.77, and 0.85, respectively. AI-ECG predicted age correlated highly with actual patient age (Spearman rho = 0.59, p < 2 x 10 -16). Amyloid risk scores were highly correlated with a known clinical history of amyloidosis (median 0.45 versus 0.05, Wilcoxon rank-sum p < 0.001). Patients with EF < 50% had greater prediction scores for cardiomyopathy compared to patients with normal EF (median 0.23 versus 0.01, Wilcoxon rank-sum p <0.001). In univariable analysis, increasing AI-ECG predicted age (HR 1.02, CI: 1.003 - 1.04, p = 0.02) and predicted AF risk score (HR: 3.2, CI: 1.4 - 7.2, p = 0.006) were associated with risk for death, whereas predicted amyloid score and predicted low EF were not associated with decreased survival. In multivariable analysis of all patients that considered patient age, hematopoietic comorbidity index, and remission status at HCT, increasing predicted AF score trended towards association with increased risk for death (HR: 2.3, 95%CI: 1.0 - 5.5, p = 0.059). Predicted AF score correlated highly with the HCT-CI and patient age. Finally, we examined whether risk prediction for low EF correlated with peri-HCT fluid overload (FO) defined by absolute weight gain. The incidence of ≥ 10% weight gain (grade 2 FO) and ≥ 20% weight gain (grade 3 FO) at 180 days post HCT was 7.4% (95% CI: 5.8%, 9.3%) and 1.0% (95% CI: 0.5%, 1.8%), respectively. Predicted low EF was not associated with development of either grade 2 or grade 3 FO in this population. In summary, AI-ECG predicts episodes of AF in patients undergoing auto HCT for PCD and may augment transplant risk appraisal when considered in conjunction with other measures of comorbidity.
Disclosures
Shaffer:Gamida Cell: Consultancy, Research Funding; Hansa Biopharma: Consultancy. Attia:Anumana: Patents & Royalties. Lahoud:MorphoSys Inc, Kite: Consultancy. Lopez-Jimenez:Anumana: Patents & Royalties. Giralt:Amgen, Actinuum, Celgene/BMS, Kite Pharma, Janssen, Jazz Pharmaceuticals, Johnson & Johnson, Novartis, Spectrum Pharma, Takeda: Membership on an entity's Board of Directors or advisory committees; Amgen, Actinuum, Celgene/BMS, Omeros, Johnson & Johnson, Miltenyi, Takeda: Research Funding. Shah:Amgen: Research Funding; Janssen: Research Funding; BMS: Research Funding; ArcellX: Other: DSMB; Beyond Spring: Research Funding. Landau:Karyopharm, Pfizer, Juno, Prothena, Caelum Biosiences, Legend Biotech, Takeda, Janssen, Nexcella: Honoraria; Alexion Pharmaceuticals, Takeda, Janssen, Prothena, Protego: Research Funding. Usmani:Oncopeptides: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Moderna: Membership on an entity's Board of Directors or advisory committees; Merck: Research Funding; Takeda: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Membership on an entity's Board of Directors or advisory committees; Gilead Sciences: Membership on an entity's Board of Directors or advisory committees, Research Funding; K36 Therapeutics: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Membership on an entity's Board of Directors or advisory committees; GSK: Membership on an entity's Board of Directors or advisory committees, Research Funding; EdoPharma: Membership on an entity's Board of Directors or advisory committees; Array Biopharma: Research Funding; SecuraBio: Membership on an entity's Board of Directors or advisory committees; SkylineDX: Membership on an entity's Board of Directors or advisory committees, Research Funding; TeneoBio: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Meyer Squibb: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Membership on an entity's Board of Directors or advisory committees, Research Funding. Friedman:Anumana: Patents & Royalties. Noseworthy:Anumana: Patents & Royalties.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal